Abstract
Introduction
The recombinant zoster vaccine (RZV) is effective in preventing herpes zoster reactivation in the general population. We previously showed that patients with chronic lymphocytic leukemia (CLL), particularly those receiving Bruton tyrosine kinase inhibitors (BTKis), have decreased humoral immune responses following vaccination. The impact of vaccination on cellular immune responses in CLL patients is not well characterized. Understanding the effect of humoral and cellular immunity in CLL patients who are treatment naïve or receiving BTKis can inform vaccination strategies in this immunosuppressed patient population.
Methods
In this phase II open-label study (NCT03702231), patients with CLL who were either treatment naïve (TN) or receiving a BTKi (ibrutinib or acalabrutinib) received 2 doses of RZV via intramuscular injection at baseline and 3 months. Subjects were followed for 6 months and assessed for serologic response at 3 and 6 months. Serologic response was defined as a ≥ four-fold rise in anti-glycoprotein E (anti-gE) IgG serum titer at the 6 month timepoint. Cellular immune response was assessed by intracellular cytokine staining and flow cytometric analysis of gE-specific CD4+ T cells expressing upregulation of ≥2 effector molecules (interferon-γ, interleukin-2, tumor necrosis factor-α, and/or CD40 ligand). Cellular response was defined as ≥ two-fold rise over baseline and ≥320 net gE-specific CD4(2+) cells per million CD4+ T cells. Descriptive statistics were used to report vaccine response rates. Mann-Whitney test and Fisher's exact test were used to compare titers and response rates between different groups. Spearman r was used to measure the correlation between vaccine responses and clinical characteristics. All subjects completed an adverse event (AE) diary documenting any local (injection site) or systemic AE that started within 7 days after receiving the first and second vaccine dose.
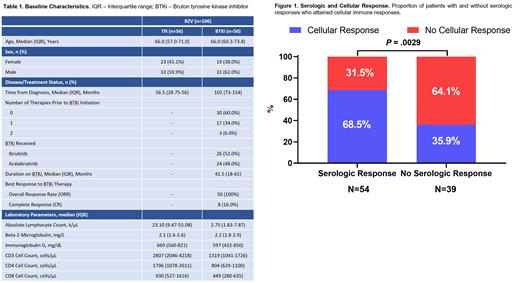
Results
106 subjects had serologic response assessment at 6 months. Baseline characteristics are shown in Table 1. The serologic response rate to RZV was significantly higher in the TN cohort (76.8%, 95% CI, 64.2-85.9; n = 56) compared to patients receiving a BTKi (40.0%, 95% CI,27.6-53.8; n = 50; P = .0002). Cellular vaccine response was assessed in 94 subjects at 6 months. Similarly, the rate of cellular immunity was significantly higher in the TN cohort (69.4%, 95% CI,55.5-80.5; n = 49) compared to patients treated with a BTKi (40.0%, 95% CI,27.0-54.5; n = 45, P = .0067). Paired serologic and cellular responses were available in 93 subjects. 68.5% (95% CI,55.3-79.3; n = 54) of subjects with a serologic response also had a positive cellular immune response, whereas 35.9% (95% CI,22.7-51.6; n = 39) of subjects attained a cellular immune response in absence of a serologic response (P = .0029) (Figure 1). Among subjects with a negative serologic response and a positive cellular immune response, 42.9% were TN (n = 6) and 57.1% (n = 8) received a BTKi. There was no difference in serologic or cellular responses between patients treated with ibrutinib and acalabrutinib (P > 0.05). Serologic antibody titers and T cell responder frequencies were weakly positively correlated (r = 0.26; 95%CI .05-.44; P = .0127). Serologic titers and T cell responses were not correlated with age, beta-2 microglobulin, absolute lymphocyte count, absolute peripheral blood CD19+, CD3+, CD4+ or CD8+ counts or serum immunoglobulin levels (IgA, IgG, IgM) (all P > 0.05). The most frequent local and systemic AEs were injection site pain (98.3%), injection site reaction (97.4%), headache (51.7%), and generalized myalgias (51.7%). Most AEs were grade 1-2 and all AEs resolved or returned to baseline within 7 days of vaccine administration.
Conclusions
RZV is safe in CLL patients and can induce both humoral and cellular immune responses. BTKi treatment was associated with impaired serologic and cellular vaccine responses compared to TN patients. Although BTKi therapy may inherently decrease vaccine immunogenicity, TN CLL patients could be more immunocompetent because of less advanced disease, thereby permitting more effective immune responses. The majority of patients with a positive antibody response also developed virus-specific T cells following vaccination. Approximately one third of patients without a positive serologic response developed virus reactive T cells.
Laing: Curevo Vaccine: Consultancy; MaxHealth LLC: Consultancy. Wiestner: Acerta Pharma: Research Funding; Pharmacyclics LLC: Research Funding; Merck: Research Funding; Nurix: Research Funding; Verastem: Research Funding; Genmab: Research Funding. Koelle: Merck: Research Funding; Curevo Vaccine: Other: Scientific Advisory Board ; MaxHealth LLC: Other: Scientific Advisory Board ; Oxford Immunotec: Research Funding; Sensei Biotherapeutics: Research Funding; Sanofi Pasteur: Research Funding. Sun: Genmab: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal